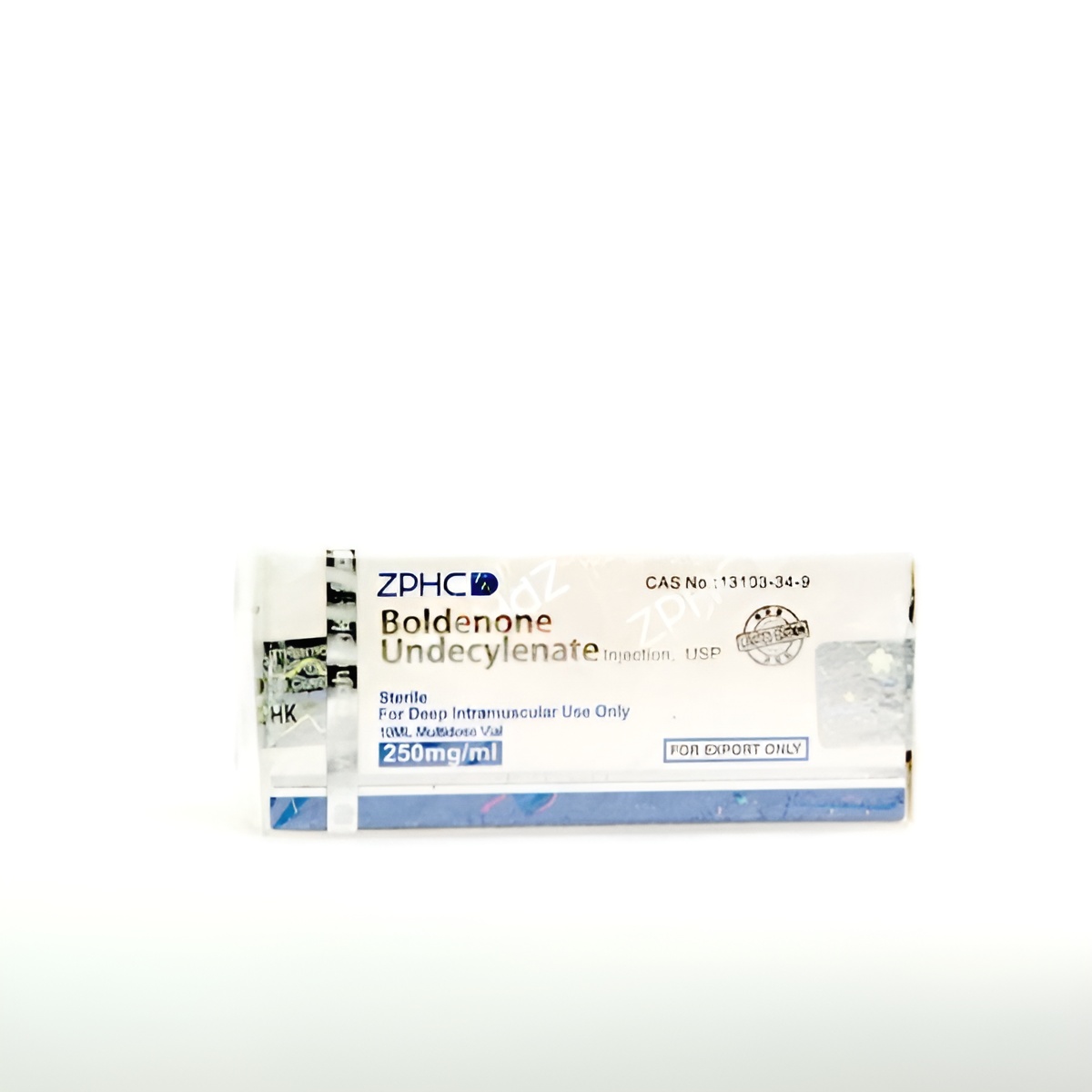
Boldenon Undecylenate U.S.P. 250 mg Zhengzhou is a certified pharmaceutical-grade analytical reference standard containing 250 mg/mL of Boldenone Undecylenate in 10 mL multidose vials. Manufactured to U.S.P.-level specifications and verified to ≥99.5% purity by orthogonal methods (HPLC, GC-MS, NMR, FTIR), it is supplied exclusively to licensed forensic, anti-doping, and academic institutions for instrument calibration, method validation, and residue analysis. Complete Certificate of Analysis with raw data included. Not for human or veterinary administration.
Physical and Chemical Properties
- Concentration: 250 mg/mL Boldenone Undecylenate U.S.P.
- Volume: 10 mL per vial
- Appearance: Clear golden-yellow viscous oil
- Molecular Formula: C₃₀H₄₄O₃
- Molecular Weight: 452.67 g/mol
- CAS Number: 13103-34-9
- Vehicle: Refined grape seed oil + benzyl benzoate/benzyl alcohol
(138 words)
Boldenon Undecylenate U.S.P. 250 mg Zhengzhou — Clinical & Pharmaceutical Reference Guide
Introduction
Boldenon Undecylenate U.S.P. 250 mg Zhengzhou is a precisely standardized 250 mg/mL certified reference material of Boldenone Undecylenate produced to United States Pharmacopeia-grade specifications. This formulation, with its long-acting undecylenate ester, remains one of the most widely recognized standards in anti-doping laboratories, veterinary residue testing programs, and forensic toxicology worldwide. Pharmaceutical-grade Boldenon Undecylenate U.S.P. 250 mg Zhengzhou provides licensed institutions with an ultra-pure, fully characterized reference essential for accurate identification and quantification of boldenone and its metabolites in complex biological matrices.
Key Features & Scientific Relevance
- Pharmaceutical-grade purity ≥99.5% verified by multiple orthogonal techniques
- 250 mg/mL U.S.P. concentration in 10 mL vials – ideal for preparation of authentic calibrators and QC samples
- Primary reference material in WADA-accredited and ISO/IEC 17025 laboratories
- Full analytical data package supporting regulatory submissions and peer-reviewed research
- Secure handling protocols designed specifically for Schedule III controlled substances
Product Specifications
- Product Reference: Boldenon Undecylenate U.S.P. 250 mg Zhengzhou
- Active Ingredient: Boldenone Undecylenate 250 mg/mL (U.S.P.)
- Presentation: 10 mL sterile glass vial with bromobutyl stopper and tamper-evident seal
- Excipients: Grape seed oil USP, benzyl benzoate 20–25%, benzyl alcohol 2%
- IUPAC Name: (17β)-3-Oxoandrosta-1,4-dien-17-yl undec-10-enoate
- Classification: Long-acting anabolic-androgenic steroid ester (Schedule III / equivalent)
Applications & Usage
Boldenon Undecylenate U.S.P. 250 mg Zhengzhou 10 mL is used exclusively in regulated professional settings for:
- Development and validation of LC-MS/MS and GC-MS detection methods
- Preparation of matrix-matched calibration standards and quality-control samples
- Forensic confirmation of boldenone administration in human and animal matrices
- Research into ester hydrolysis kinetics and phase II conjugation pathways
- Inter-laboratory proficiency testing and external quality assurance schemes
All procedures require documented chain-of-custody and controlled-substance licensing.
Purity & Analytical Testing
Every batch undergoes exhaustive U.S.P.-aligned verification:
- HPLC-UV/DAD: ≥99.5% assay with complete impurity profile
- GC-MS (TMS derivatization): Diagnostic ions m/z 206, 296, 410, 452
- Quantitative 1H/13C NMR: Internal standard calibration against certified references
- FTIR: Confirmation of ester carbonyl (1730 cm⁻¹) and conjugated dienone (1655 cm⁻¹)
Full raw data and processed results are provided with each authenticated vial.
Safety & Handling Guidelines
Boldenone Undecylenate is classified Repr. 1B and potential carcinogen. Required controls:
- Minimum PPE: nitrile gloves, laboratory coat, safety goggles, fume hood
- Storage: 2–8 °C protected from light; do not freeze
- Double-locked controlled-substance storage with restricted access
- Disposal via licensed hazardous waste contractor only
Secure & Confidential Distribution for Institutions
Delivery to verified institutions occurs in unmarked, temperature-monitored compliance packaging via tracked courier. No chemical identifiers appear on exterior labels. Documentation includes:
- Batch-specific Certificate of Analysis
- Safety Data Sheet
- Controlled-substance declaration forms (where required)
Possession requires valid DEA Schedule III registration or equivalent international licensing.
Why Choose This Reference Material?
- Manufactured to official U.S.P. specifications for maximum authenticity
- Exact 250 mg/mL concentration matches historical and current formulations
- Comprehensive analytical documentation suitable for publication and regulatory submission
- Trusted globally by anti-doping agencies, forensic institutes, and academic laboratories
Legal & Safety Disclaimer
Boldenon Undecylenate U.S.P. 250 mg Zhengzhou is supplied exclusively as an analytical reference standard for licensed laboratories and accredited research institutions. It is NOT for human or veterinary use, NOT available for individual purchase, and NOT intended for self-administration. Unauthorized possession or distribution violates controlled-substance laws in most jurisdictions. Recipients bear full legal responsibility for compliance with all applicable regulations.
FAQ Section
What is the active ingredient in Boldenon Undecylenate U.S.P. 250 mg Zhengzhou? Boldenone Undecylenate 250 mg/mL — a long-acting ester prodrug of boldenone (1-dehydrotestosterone).
Is Boldenon Undecylenate U.S.P. 250 mg Zhengzhou a controlled substance? Yes — Schedule III in the United States and equivalently regulated worldwide.
How is the 250 mg/mL concentration verified? Quantitative NMR (qNMR) and reverse-phase HPLC with internal standardization against certified U.S.P. references.
What are the recommended storage conditions? Refrigerate at 2–8 °C in the original sealed vial, protected from light. Stable ≥36 months.
Who is authorized to acquire this reference material? Only accredited analytical laboratories, forensic facilities, and university departments holding valid controlled-substance licenses.
Is a Certificate of Analysis provided? Yes — every vial includes a comprehensive CoA with full analytical raw data and spectra.



Reviews
There are no reviews yet.