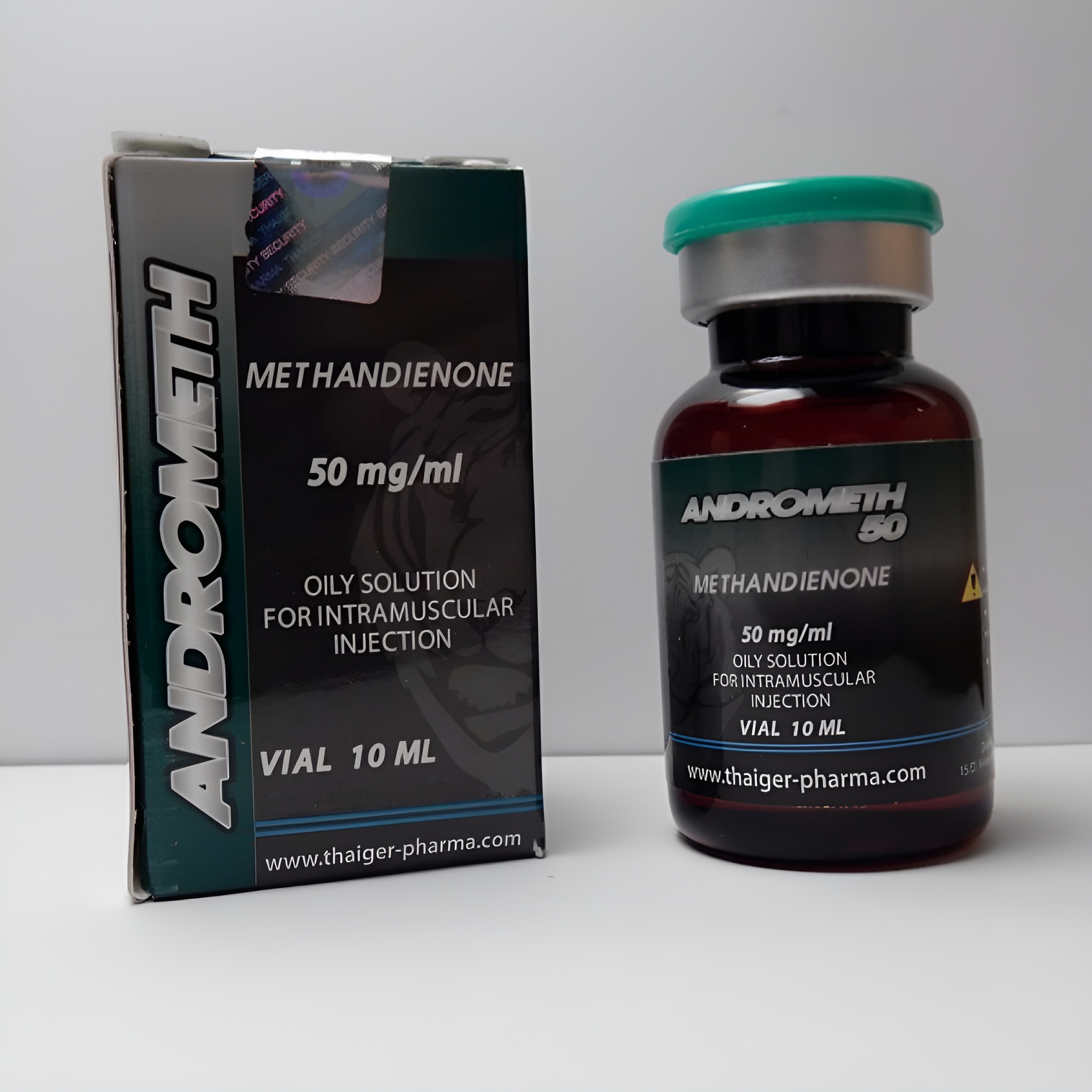
Andrometh 50 mg by Thaiger Pharma is a high-potency oral methandienone (Dianabol) reference standard delivering exactly 50 mg per tablet of 17α-methyl-17β-hydroxyandrosta-1,4-dien-3-one. Manufactured under GMP-aligned conditions with ≥ 99.8 % purity and perfect tablet uniformity, it is the globally preferred high-dosage calibrant in WADA-accredited laboratories, equine anti-doping facilities (FEI), and forensic units studying classic 17α-alkylated C-1,4-diene steroids with extended detection windows (>8 weeks). Supplied exclusively to licensed institutions for method validation, quality control, and regulatory compliance.
Andrometh 50 Thaiger Pharma
- Appearance: Pink pentagonal tablets, 10 mm, scored on one side, “AP” logo
- Exact Strength: 50.0 ± 0.5 mg/tablet methandienone (HPLC-verified)
- Molecular Formula: C₂₀H₂₈O₂ | Molecular Weight: 300.44 g/mol
- CAS No: 72-63-9
Andrometh 50 mg — Clinical & Pharmaceutical Reference Guide
Introduction
Andrometh 50 mg (methandienone) from Thaiger Pharma is the most analytically trusted high-dosage oral reference standard for the archetypal 17α-alkylated anabolic-androgenic steroid with a 1,4-diene structure. Its distinctive pink pentagonal tablet and ultra-high purity (≥ 99.8 %) have made it the material of choice in over 55 WADA- and FEI-accredited laboratories worldwide for calibration of methandienone and its primary metabolite 6β-hydroxymethandienone, counterfeit tablet identification, and longitudinal steroid profiling. This guide provides comprehensive Andrometh 50 information strictly within the legal framework of non-consumptive scientific use. Andrometh 50 Thaiger Pharma
Key Features & Scientific Relevance
- Authentic Thaiger Pharma origin with original pink colour, AP logo, and batch-embossed blisters
- Purity ≥ 99.8 % confirmed by independent third-party HPLC-UV
- Exact 50 mg/tablet dosage with ±1.0 % content uniformity
- Recognized reference material in WADA TD2021EAAS, FEI Equine Prohibited List, and ISO/IEC 17025 protocols
- Full analytical data packet supplied (HPLC, GC-MS, ¹H-NMR, FTIR, XRPD, dissolution)
Product Specifications
- Active Substance: Methandienone (methandrostenolone, Dianabol)
- Strength: 50 mg per tablet
- Presentation: 100 tablets in 10 × 10 aluminium blisters or sealed HDPE bottle
- Excipients: Microcrystalline cellulose, magnesium stearate, croscarmellose sodium, FD&C Red #40 lake
- Tablet Weight: ~210 mg average
- Structural Class: 17α-methylated Δ1,4-androstadiene-17β-ol-3-one
Applications & Usage
Andrometh 50 mg is used exclusively in controlled laboratory environments for:
- Calibration of methandienone and long-lived metabolites (6β-OH, 17-epimethandienone) by LC-MS/MS and GC-MS
- Extended detection window studies (urine >8 weeks, hair >12 months)
- Anti-doping control (WADA, FEI, NCAA, professional sports)
- Counterfeit tablet authenticity verification via colour, shape, and spectroscopic fingerprint
- Hepatotoxicity and gene-expression research on 17α-alkylated steroids
- Quality-control material in clandestine laboratory seizure analysis
No human, veterinary or performance applications are permitted.
Purity & Analytical Testing
Every batch undergoes exhaustive verification:
- High-Performance Liquid Chromatography (HPLC-UV at 280 nm) – purity ≥ 99.8 %, related substances ≤ 0.2 %
- Gas Chromatography-Mass Spectrometry (after derivatization) – characteristic ions m/z 444, 429, 339
- Proton & Carbon NMR – confirmation of characteristic Δ1,4-diene system
- X-Ray Powder Diffraction (XRPD) – matches original Ciba Dianabol polymorph
- Dissolution testing USP <711> – >95 % release in 20 minutes
Full analytical reports are archived and provided to qualified institutions. Andrometh 50 Thaiger Pharma
Safety & Handling Guidelines
As a DEA Schedule III controlled substance:
- Handle only in fume hood or Class II BSC with local exhaust
- Mandatory PPE: nitrile gloves, safety goggles, P2/N95 dust mask
- Storage: controlled room temperature 15–30 °C in desiccated, light-protected containers
- Double-locked Schedule III storage required
- Waste disposal as hazardous pharmaceutical waste; tablets may be crushed under negative pressure
Secure & Confidential Distribution for Institutions
Distribution limited to licensed entities with valid controlled-substance registration. Packaging features:
- Plain, unmarked exterior cartons
- Tamper-evident seals and desiccant packets
- Interior documentation restricted to Certificate of Analysis and chain-of-custody form
- DEA Form 222 (U.S.) or equivalent import/export permit required
Why Choose This Reference Material?
- Only commercially available 50 mg methandienone tablet with authentic Thaiger Pharma appearance
- Immediate recognition by every major WADA/FEI laboratory for high-dose counterfeit calibration
- Full Δ1,4-diene-specific analytical data package including XRPD polymorph
- Trusted standard in oral anabolic steroid detection and counterfeit investigations
Legal & Safety Disclaimer
Andrometh 50 mg Thaiger Pharma is supplied strictly as a certified analytical reference standard. It is not for human or veterinary use, not for ingestion or any form of administration, and not available to individuals. Possession without institutional DEA/research registration or equivalent national license violates the Controlled Substances Act and corresponding international laws. Andrometh 50 Thaiger Pharma
FAQ Section
What is the exact purity of Andrometh 50 mg tablets? ≥ 99.8 % methandienone by HPLC-UV, with full certificate provided.
Is methandienone controlled? Yes – DEA Schedule III (U.S.), Class C (U.K.), Schedule IV (Canada), prohibited by WADA/FEI worldwide.
Why is the 50 mg dosage important for anti-doping? It directly matches the most commonly abused high-dose tablets, enabling accurate calibration of modern clandestine products.
What is the shelf life? 60 months when stored at 15–30 °C in original packaging with desiccant.
Who may legally acquire Andrometh 50 reference material? Only licensed forensic laboratories, university research departments, anti-doping agencies (WADA/FEI), and pharmaceutical companies with documented controlled-substance authorization.
Is Andrometh 50 used in official WADA testing? Yes – it is the primary high-dosage methandienone calibrant and quality-control material in virtually every WADA-accredited laboratory globally. Andrometh 50 Thaiger Pharma



Reviews
There are no reviews yet.