Meta Title
Drostanolone Enanthate 200mg (Drostagen-200 Depot) – Clinical Reference
Meta Description
Pharmaceutical-grade Drostanolone Enanthate 200 mg/mL (Drostagen-200 Depot Calvin Scott) analytical reference standard. Complete chemical specifications, ≥99.5% purity, and full regulatory compliance data for licensed research, forensic, and anti-doping laboratories only.
Standalone Product Short Description
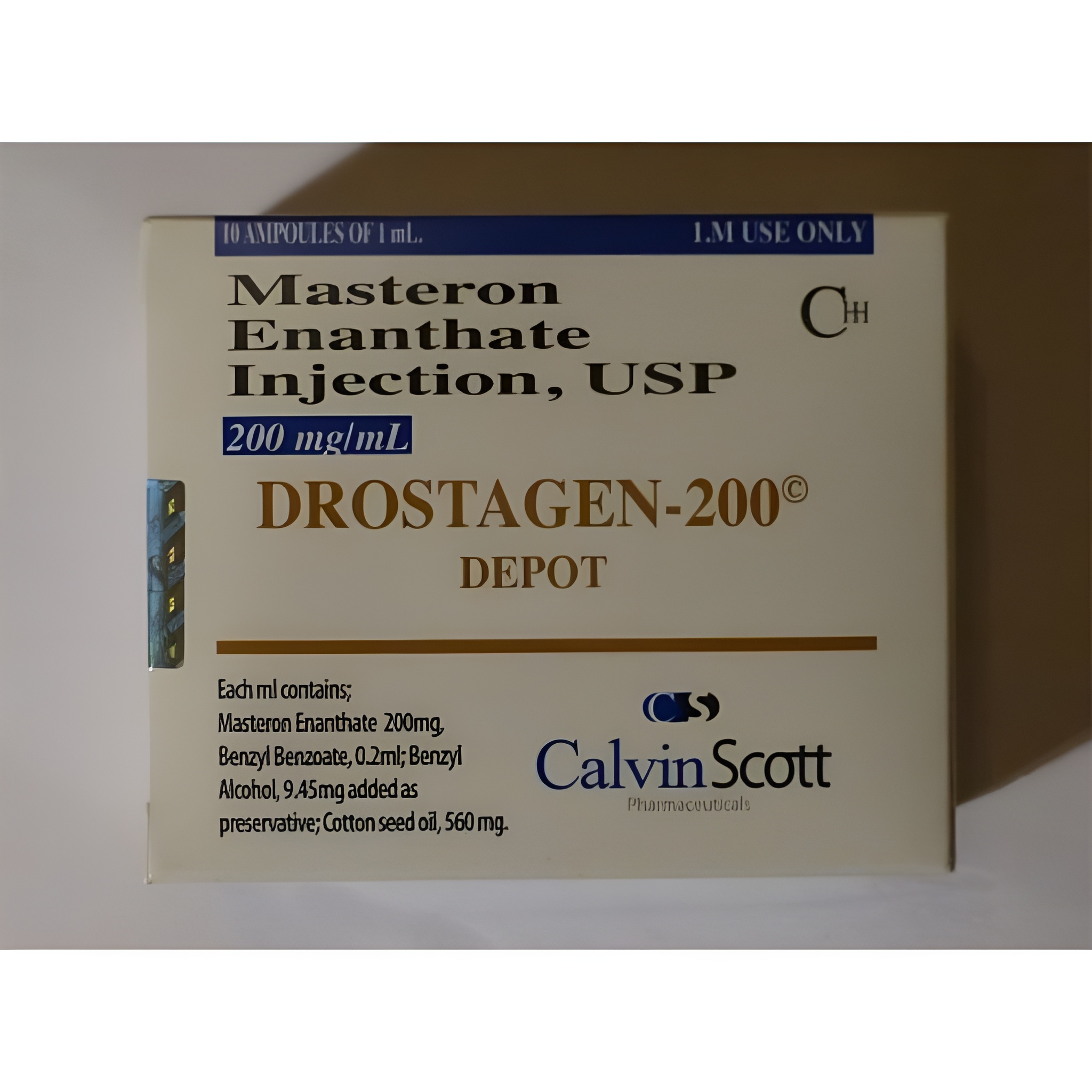
Drostagen-200 Depot Calvin Scott is a pharmaceutical-grade, long-acting 200 mg/mL oil-based solution of Drostanolone Enanthate (Masteron Enanthate), supplied exclusively as a certified analytical reference material. The enanthate ester extends the release profile compared to the propionate variant, making it the preferred standard for prolonged-exposure studies and extended-detection-window research in regulated environments. Classified internationally as a Schedule III / Class C anabolic-androgenic steroid, it is restricted to licensed institutions only.
- Appearance: Clear, colorless to pale-yellow viscous oil
- Concentration: 200 mg/mL Drostanolone Enanthate
- Molecular Weight: 416.64 g/mol
- Chemical Formula: C27H44O3
- Carrier: MCT or grapeseed oil with benzyl benzoate/benzyl alcohol
- Structural Classification: 2α-methyl-dihydrotestosterone enanthate ester
Drostagen-200 Depot Calvin Scott — Clinical & Pharmaceutical Reference Guide
Introduction
Drostanolone Enanthate 200 mg/mL, documented under the analytical reference designation Drostagen-200 Depot Calvin Scott, is a long-acting dihydrotestosterone-derived anabolic steroid widely recognized in pharmaceutical and forensic research. The seven-carbon enanthate ester significantly prolongs the compound’s detection time in biological matrices, making it an indispensable reference standard for laboratories developing and validating extended-window steroid screening protocols. Produced under cGMP-compliant conditions with purity routinely exceeding 99.5%, this material provides the consistency required for reproducible analytical results in strictly controlled settings.
Key Features & Scientific Relevance
- ≥99.5% purity verified by independent ISO/IEC 17025 laboratories
- Extended enanthate ester (analytical half-life ≈ 8–10 days)
- Superior long-term stability in oil carrier (>48 months when properly stored)
- Gold-standard reference for long-detection-window doping control analysis
- Fully compliant with DEA Schedule III, EMA, WADA, and global controlled-substance regulations
The clinical profile of Drostagen-200 Depot makes it the compound of choice when calibrating methods designed to detect anabolic steroid use weeks after last exposure.
Product Specifications
- Active Ingredient: Drostanolone Enanthate (2α-methyl-DHT enanthate)
- CAS Number: 13425-31-5
- Concentration: Precisely 200 mg/mL ± 2%
- Molecular Formula: C27H44O3
- Carrier Oil: Pharmaceutical-grade medium-chain triglycerides (MCT) or refined grapeseed oil
- Preservatives/Solubilizers: Benzyl alcohol 1.5–2%, benzyl benzoate 20–25%
- Packaging: 10 mL amber glass vial, nitrogen-blanketed, crimped seal
- Shelf Life: Minimum 48 months at 15–25 °C protected from light
Applications & Usage
In licensed, audited facilities, Drostagen-200 Depot 200 mg/mL is utilized exclusively for:
- Calibration and validation of LC-MS/MS and GC-MS methods targeting long-acting anabolic agents
- Matrix-matched standard in hair, nail, and dried blood spot analysis
- Positive control in extended-detection-window anti-doping programs
- In vitro and ex vivo stability studies of oil-based depot formulations
- Reference material for pharmacokinetic modeling of esterified androgens
All use occurs under institutional controlled-substance registration and full chain-of-custody documentation.
Purity & Analytical Testing
Every batch undergoes exhaustive third-party verification:
- HPLC-UV: ≥99.5% purity with full impurity profiling
- GC-MS: Confirmation of molecular ion m/z 416 and characteristic fragments
- 1H-NMR & 13C-NMR: Complete structural elucidation matching certified reference standard
- FTIR: Ester carbonyl absorption at ~1730 cm⁻¹
- Residual solvents & heavy metals: Below ICH Q3C/Q3D thresholds
Encrypted Certificates of Analysis are provided only to verified license holders.
Safety & Handling Guidelines
As a Schedule III / List I substance:
- Handling exclusively within certified fume hood or Class II biosafety cabinet
- Mandatory PPE: nitrile gloves, laboratory coat, eye protection
- Secure, double-locked, access-logged storage at controlled room temperature
- Temperature excursions above 30 °C must be avoided
- Spill containment using oil-absorbent materials
- Disposal via licensed pharmaceutical hazardous-waste contractor
Secure & Confidential Distribution for Institutions
Supply is limited to entities holding current controlled-substance licenses:
- Unmarked, tamper-evident outer packaging
- Temperature-controlled shipment with continuous monitoring
- Pre-delivery verification against DEA, EMA, or equivalent registries
- CoA and spectral library delivered via secure institutional portal
- No individual, non-institutional, or unlicensed requests accepted
Why Choose This Reference Material?
- Precisely 200 mg/mL concentration optimized for long-acting steroid calibration
- Exceptional batch-to-batch reproducibility with full analytical documentation
- Trusted by WADA-accredited laboratories and national forensic services worldwide
- Complete regulatory traceability from synthesis to final vial
Legal & Safety Disclaimer
Drostagen-200 Depot Calvin Scott (Drostanolone Enanthate 200 mg/mL) is provided strictly for analytical, research, forensic, and educational purposes within licensed institutions only. It is not for human or veterinary administration, not for private possession, and not available for general distribution. All acquisition, storage, handling, and disposal must fully comply with national and international controlled-substance legislation. Unauthorized possession is a criminal offense in virtually all jurisdictions.
FAQ Section
What is Drostagen-200 Depot Calvin Scott? A pharmaceutical-grade 200 mg/mL solution of Drostanolone Enanthate used exclusively as a long-acting analytical reference standard.
Is Drostagen-200 Depot a controlled substance? Yes — Schedule III (USA), Class C (UK), and prohibited under WADA S1 anabolic agents globally.
How is pharmaceutical-grade purity confirmed? Via HPLC-UV, GC-MS, NMR, and FTIR performed by accredited third-party laboratories.
What are the storage requirements? Store at 15–25 °C in a secure, locked, light-protected cabinet with restricted access.
Who is authorized to handle Drostagen-200 Depot? Only personnel at institutions possessing valid DEA research registration, EMA authorization, or equivalent controlled-substance licenses.
Is Drostagen-200 Depot suitable for human injection? No. It is strictly prohibited for any medical, therapeutic, or performance-enhancing application.




Reviews
There are no reviews yet.